Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Sugita, Tsuyoshi; Kobayashi, Kentaro*; Yamazaki, Taiki*; Isaka, Mayu*; Itabashi, Hideyuki*; Mori, Masanobu*
Journal of Photochemistry and Photobiology A; Chemistry, 400, p.112662_1 - 112662_8, 2020/09
Times Cited Count:1 Percentile:2.52(Chemistry, Physical)In this study, we developed an in-line photocatalytic performance evaluation system in which a flow reactor was connected to the ion chromatography to accurately evaluate the performance of the photocatalyst. This system was used to evaluate the photocatalyst supported by the two-layer support method on the substrate, such as glass beads. The performance of the photocatalyst was evaluated using dimethyl sulfoxide (DMSO), and it was possible to monitor the decomposition of DMSO by UV and the formation of by-products, such as methane sulfonate (MSO) and sulfate (SA). This system can be expected to be useful not only for evaluating the decomposition performance of an object using a photocatalyst but also for evaluating the byproducts.
 films by RBS/C
films by RBS/CYamamoto, Shunya; Takeyama, Akinori; Yoshikawa, Masahito
Nuclear Instruments and Methods in Physics Research B, 242(1-2), p.377 - 379, 2006/01
Times Cited Count:9 Percentile:53.56(Instruments & Instrumentation)no abstracts in English
 (100) thin films by helium ion implantation
(100) thin films by helium ion implantationYamamoto, Shunya; Nagata, Shinji*; Takeyama, Akinori; Yoshikawa, Masahito
Transactions of the Materials Research Society of Japan, 30(3), p.789 - 792, 2005/09
no abstracts in English
 /ZnO films by pulsed laser deposition
/ZnO films by pulsed laser depositionYamamoto, Shunya; Choi, Y.; Umebayashi, Tsutomu; Yoshikawa, Masahito
Transactions of the Materials Research Society of Japan, 29(6), p.2701 - 2704, 2004/00
no abstracts in English
 under visibel light irradiation
under visibel light irradiationUmebayashi, Tsutomu; Asai, Keisuke*; Yamaki, Tetsuya; Yoshikawa, Masahito
Kogyo Zairyo, 51(7), p.34 - 36, 2003/07
no abstracts in English
 by ion implantation
by ion implantationUmebayashi, Tsutomu; Yamaki, Tetsuya; Yamamoto, Shunya; Tanaka, Shigeru; Asai, Keisuke*
Transactions of the Materials Research Society of Japan, 28(2), p.461 - 464, 2003/06
TiO is promising as a photocatalytic material. However, it is active only under UV light irradiation because of its wide band gap (
is promising as a photocatalytic material. However, it is active only under UV light irradiation because of its wide band gap ( 3.0 eV). We recently reported that sulfur (S) doping caused the optical absorption edge of TiO
3.0 eV). We recently reported that sulfur (S) doping caused the optical absorption edge of TiO to be shifted into the lower energy region. Based on the theoretical analyses using first principles band calculations, mixing of the S 3p states with the valence band was found to contribute to the bandgap narrowing. In this study presented here, S-doped TiO
to be shifted into the lower energy region. Based on the theoretical analyses using first principles band calculations, mixing of the S 3p states with the valence band was found to contribute to the bandgap narrowing. In this study presented here, S-doped TiO was prepared by ion implantation and subsequent thermal annealing. S
was prepared by ion implantation and subsequent thermal annealing. S was implanted into the single crystals of rutile TiO
was implanted into the single crystals of rutile TiO with a fluence of 8
with a fluence of 8  10
10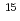 ions/cm
ions/cm . According to the results of RBS/channeling analysis, irradiation damage recovered after the annealing at 600
. According to the results of RBS/channeling analysis, irradiation damage recovered after the annealing at 600  C in air. In the annealed crystal, S atoms occupied O sites to form Ti-S bonds, as assessed by XPS.
C in air. In the annealed crystal, S atoms occupied O sites to form Ti-S bonds, as assessed by XPS.
Umebayashi, Tsutomu; Yamaki, Tetsuya; Yamamoto, Shunya; Miyashita, Atsumi; Tanaka, Shigeru; Sumita, Taishi*; Asai, Keisuke*
Journal of Applied Physics, 93(9), p.5156 - 5160, 2003/05
Times Cited Count:300 Percentile:98.62(Physics, Applied)Sulfur (S)-doped titanium dioxide (TiO ) was synthesized by ion implantation and subsequent thermal annealing. Compared to the pure TiO
) was synthesized by ion implantation and subsequent thermal annealing. Compared to the pure TiO , a photocurrent was observed in the lower energy regions for the S-doped TiO
, a photocurrent was observed in the lower energy regions for the S-doped TiO . Based on the theoretical analyses by the first principles band calculations using the full potential linearized augmented plane wave methods within the generalized gradient approximation, the mixing of the S 3p states with the valence band (VB) was found to contribute to the increasing width of the VB. This leads to the bandgap narrowing in the S-doped TiO
. Based on the theoretical analyses by the first principles band calculations using the full potential linearized augmented plane wave methods within the generalized gradient approximation, the mixing of the S 3p states with the valence band (VB) was found to contribute to the increasing width of the VB. This leads to the bandgap narrowing in the S-doped TiO . Therefore, the photon-to-carrier conversion was induced during irradiation by visible light above 420 nm (
. Therefore, the photon-to-carrier conversion was induced during irradiation by visible light above 420 nm ( 2.9 eV).
2.9 eV).
 films by RBS/channeling
films by RBS/channelingYamamoto, Shunya; Yamaki, Tetsuya; Naramoto, Hiroshi; Tanaka, Shigeru
Nuclear Instruments and Methods in Physics Research B, 206(1-4), p.268 - 271, 2003/05
Times Cited Count:13 Percentile:64.66(Instruments & Instrumentation)no abstracts in English

Umebayashi, Tsutomu; Yamaki, Tetsuya; Tanaka, Shigeru; Asai, Keisuke*
Chemistry Letters, 32(4), p.330 - 331, 2003/04
Times Cited Count:501 Percentile:99.17(Chemistry, Multidisciplinary)A sulfur (S)-doped titanium dioxide (TiO ) photocatalyst was synthesized by oxidative annealing of titanium disulfide (TiS
) photocatalyst was synthesized by oxidative annealing of titanium disulfide (TiS ). TiS
). TiS turned into anatase TiO
turned into anatase TiO when annealed in air. The residual S atoms occupied O-atom sites in the TiO
when annealed in air. The residual S atoms occupied O-atom sites in the TiO to form Ti-S bonds. The S doping caused the absorption edge of TiO
to form Ti-S bonds. The S doping caused the absorption edge of TiO to be shifted into the lower energy region. Consequently, methylene blue adsorbed on the S-doped TiO
to be shifted into the lower energy region. Consequently, methylene blue adsorbed on the S-doped TiO was photocatalytic decomposed by visible light irradiation.
was photocatalytic decomposed by visible light irradiation.
 based on band calculations
based on band calculationsUmebayashi, Tsutomu; Yamaki, Tetsuya; Ito, Hisayoshi; Asai, Keisuke*
Journal of Physics and Chemistry of Solids, 63(10), p.1909 - 1920, 2002/10
Times Cited Count:627 Percentile:99.67(Chemistry, Multidisciplinary)The electronic structures of titanium dioxide (TiO ) doped with 3d transition metals (V, Cr, Mn, Fe, Co and Ni) have been analyzed by ab-initio band calculations based on the density functional theory with the full-potential linearized-augmented-plane-wave methods. When TiO
) doped with 3d transition metals (V, Cr, Mn, Fe, Co and Ni) have been analyzed by ab-initio band calculations based on the density functional theory with the full-potential linearized-augmented-plane-wave methods. When TiO is doped with V, Cr, Mn, Fe or Co, an occupied level occurs and the electrons localized around each dopant. As the atomic number of the dopant increases, the localized level shifts to lower energy. In contrast, the electrons from Ni dopant are somewhat delocalized, thus significantly contributing to the formation of the valence band with O p and Ti 3d electrons. Based on a comparison with the absorption and photoconductivity data previously reported, we show that the t
is doped with V, Cr, Mn, Fe or Co, an occupied level occurs and the electrons localized around each dopant. As the atomic number of the dopant increases, the localized level shifts to lower energy. In contrast, the electrons from Ni dopant are somewhat delocalized, thus significantly contributing to the formation of the valence band with O p and Ti 3d electrons. Based on a comparison with the absorption and photoconductivity data previously reported, we show that the t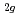 state of the dopant plays a significant role in the photoresponse of TiO
state of the dopant plays a significant role in the photoresponse of TiO under visible light irradiation.
under visible light irradiation.
Umebayashi, Tsutomu; Yamaki, Tetsuya; Ito, Hisayoshi; Asai, Keisuke*
Applied Physics Letters, 81(3), p.454 - 456, 2002/07
Times Cited Count:1350 Percentile:99.93(Physics, Applied)Titanium dioxide (TiO ) doped with sulfur (S) was synthesized by oxidation annealing of titanium disulfide (TiS
) doped with sulfur (S) was synthesized by oxidation annealing of titanium disulfide (TiS ). According to the X-ray diffraction patterns, TiS
). According to the X-ray diffraction patterns, TiS turned into anatase TiO
turned into anatase TiO when annealed at 600
when annealed at 600  C. The residual S atoms occupied O-atom sites in TiO
C. The residual S atoms occupied O-atom sites in TiO to form Ti-S bonds. The S doping caused the absorption edge of TiO
to form Ti-S bonds. The S doping caused the absorption edge of TiO to be shifted into the lower energy region. Based on the theoretical analyses using ab-initio band calculations, mixing of the S 3p states with the valence band was found to contribute to the band gap narrowing.
to be shifted into the lower energy region. Based on the theoretical analyses using ab-initio band calculations, mixing of the S 3p states with the valence band was found to contribute to the band gap narrowing.
Syarif, D. G.; Miyashita, Atsumi; Yamaki, Tetsuya; Sumita, Taishi*; Choi, Y.; Ito, Hisayoshi
Applied Surface Science, 193(1-4), p.287 - 292, 2002/06
Times Cited Count:91 Percentile:93.64(Chemistry, Physical)Titanium dioxide ( ) films on glass substrates were prepared by pulsed laser deposition (PLD) technique. The dependence of crystal structure, morphology and photocatalytic activity on oxygen partial pressure were investigated using X-ray diffraction (XRD), Atomic force microscope (AFM) and UV-VIS Spectrophotometer. By increasing oxygen partial pressure, the surface morphology changed from smooth to rough, and the crystal structure changed from rutile to anatase at fixed substrate temperature of 773 K. The change of the surface morphology and the formation of anatase structure by variation of oxygen partial pressure resulted in an increase of photocatalytic activity.
) films on glass substrates were prepared by pulsed laser deposition (PLD) technique. The dependence of crystal structure, morphology and photocatalytic activity on oxygen partial pressure were investigated using X-ray diffraction (XRD), Atomic force microscope (AFM) and UV-VIS Spectrophotometer. By increasing oxygen partial pressure, the surface morphology changed from smooth to rough, and the crystal structure changed from rutile to anatase at fixed substrate temperature of 773 K. The change of the surface morphology and the formation of anatase structure by variation of oxygen partial pressure resulted in an increase of photocatalytic activity.
 films prepared by pulsed laser deposition
films prepared by pulsed laser depositionYamamoto, Shunya; Sumita, Taishi; Yamaki, Tetsuya; Miyashita, Atsumi; Naramoto, Hiroshi
Journal of Crystal Growth, 237-239(Part1), p.569 - 573, 2002/04
The epitaxial growth of TiO films has attracted much interest from the viewpoints of basic science and applications. In the present study, it is shown that TiO
films has attracted much interest from the viewpoints of basic science and applications. In the present study, it is shown that TiO films with anatase or rutile structure can be grown by pulsed laser deposition(PLD) with an ArF excimer laser under the controlled O
films with anatase or rutile structure can be grown by pulsed laser deposition(PLD) with an ArF excimer laser under the controlled O atmosphere. The TiO
atmosphere. The TiO films were prepared on the LaAlO
films were prepared on the LaAlO , LSAT SrTiO
, LSAT SrTiO and
and  -Al
-Al O
O single crystal substrates. The epitaxial films were analyzed by RBS and X-ray diffraction for investigating the crystallographic relationships with the substrates. The high quality anatase TiO
single crystal substrates. The epitaxial films were analyzed by RBS and X-ray diffraction for investigating the crystallographic relationships with the substrates. The high quality anatase TiO (001) films have been successfully grown on the LaAlO
(001) films have been successfully grown on the LaAlO (001), LSAT (001) and SrTiO
(001), LSAT (001) and SrTiO (001) substrates at temperatures above 500
(001) substrates at temperatures above 500 C. Also the high quality rutile TiO
C. Also the high quality rutile TiO (100) and (001) films were obtained on the
(100) and (001) films were obtained on the  -Al
-Al O
O (0001) and (10-10), respectively.The optical properties were characterized by optical absorption measurements. The optical band gaps for anatase and rutile TiO
(0001) and (10-10), respectively.The optical properties were characterized by optical absorption measurements. The optical band gaps for anatase and rutile TiO epitaxial films were evaluated to be 3.22 eV and 3.11 eV, respectively.
epitaxial films were evaluated to be 3.22 eV and 3.11 eV, respectively.
 films by pulsed laser deposition technique
films by pulsed laser deposition techniqueYamamoto, Shunya; Sumita, Taishi; Sugiharuto; Miyashita, Atsumi; Naramoto, Hiroshi
Thin Solid Films, 401(1-2), p.88 - 93, 2001/12
Times Cited Count:129 Percentile:96.77(Materials Science, Multidisciplinary)The epitaxial growth of high-quality TiO films has attracted much interest from the viewpoints of basic science and applications. In this study, it is shown that TiO
films has attracted much interest from the viewpoints of basic science and applications. In this study, it is shown that TiO films with anatase and rutile structure can be prepared by pulsed laser deposition (PLD) with a Nd-YAG laser under the controlled O
films with anatase and rutile structure can be prepared by pulsed laser deposition (PLD) with a Nd-YAG laser under the controlled O atmosphere. The TiO
atmosphere. The TiO films with 200 nm thickness were prepared on the SrTiO
films with 200 nm thickness were prepared on the SrTiO , LaAlO
, LaAlO , LSAT and
, LSAT and  -Al
-Al O
O single crystal substrates. The epitaxial films were analyzed by Rutherford backscattering spectrometry (RBS) combined with channeling and X-ray diffraction for the crystallographic relationships with the substrates. The high quality anatase TiO
single crystal substrates. The epitaxial films were analyzed by Rutherford backscattering spectrometry (RBS) combined with channeling and X-ray diffraction for the crystallographic relationships with the substrates. The high quality anatase TiO (001) films have been successfully prepared on the LaAlO
(001) films have been successfully prepared on the LaAlO (001), LSAT (001) and SrTiO
(001), LSAT (001) and SrTiO (001) substrates about 500
(001) substrates about 500 C substrate temperature. Also the high quality rutile TiO
C substrate temperature. Also the high quality rutile TiO (100) and TiO
(100) and TiO (001) films were obtained on the
(001) films were obtained on the  -Al
-Al O
O (0001) and (10
(0001) and (10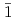 0), respectively.
0), respectively.
Sumita, Taishi; Yamaki, Tetsuya; Yamamoto, Shunya; Miyashita, Atsumi
Japanese Journal of Applied Physics, Part 1, 40(6A), p.4007 - 4008, 2001/06
Times Cited Count:8 Percentile:37.61(Physics, Applied)We propose a new simple method for characterizing photocatalytic activity by measuring photo-generated transient charge separation at the surface of semiconductor photocatalysts. In this method, the charge separation generated by a pulse dye laser is obtained as a function of the incident laser energy. Using this method, the photocatalytic activity and the type of surface reaction (reduction or oxidation) in titanium dioxide films were rapidly determined.
 single crystals by nonmetal-ion implantation
single crystals by nonmetal-ion implantationYamaki, Tetsuya; Sumita, Taishi; Yamamoto, Shunya; Miyashita, Atsumi
Dai-11-Kai Ryushisen No Sentanteki Oyo Gijutsu Ni Kansuru Shimpojiumu (BEAMS 2000) Hobunshu, p.131 - 134, 2000/11
no abstracts in English
Katsube, Daiki*; Ono, Shinya*; Takayanagi, Shuhei*; Ojima, Shoki*; Maeda, Motoyasu*; Yoshida, Hikaru*; Nishi, Shizuka*; Yoshigoe, Akitaka; Abe, Masayuki*
no journal, ,
TiO has been extensively studied because of high photocatalytic activity. However, the basic reaction process of photocatalysis has not been understood yet. In this study, the difference in water adsorption reaction between rutile and anatase was clarified using synchrotron radiation soft X-ray photoelectron spectroscopy (XPS). The OH component observed at the higher binding energy side was clearly confirmed in the rutile type TiOTiO
has been extensively studied because of high photocatalytic activity. However, the basic reaction process of photocatalysis has not been understood yet. In this study, the difference in water adsorption reaction between rutile and anatase was clarified using synchrotron radiation soft X-ray photoelectron spectroscopy (XPS). The OH component observed at the higher binding energy side was clearly confirmed in the rutile type TiOTiO , suggesting that the rutile type is more reactive to water adsorption.
, suggesting that the rutile type is more reactive to water adsorption.
Sugita, Tsuyoshi; Mori, Masanobu*; Shimoyama, Iwao
no journal, ,
This study investigates the conversion of biotite, a subgroup of clay minerals, into photocatalysts through heat treatment with CaCl . The resulting reaction products were analyzed in terms of their composition, structure, optical properties, and photocatalytic activity against Cr(VI) and salicylic acid (SA). Biotite and CaCl
. The resulting reaction products were analyzed in terms of their composition, structure, optical properties, and photocatalytic activity against Cr(VI) and salicylic acid (SA). Biotite and CaCl mixtures could be heated to 600
mixtures could be heated to 600 C while retaining the biotite crystal structure, whereas heating to 700
C while retaining the biotite crystal structure, whereas heating to 700 C resulted in transformation to octahedral wadalite crystals. The band gap of the wadalite obtained after heat treatment was approximately 3.10 eV. The photocatalytic reduction rate per unit surface area increased markedly with increasing heat-treatment temperature, and the Cr(VI) reduction and SA degradation rates of the sample calcined at 800
C resulted in transformation to octahedral wadalite crystals. The band gap of the wadalite obtained after heat treatment was approximately 3.10 eV. The photocatalytic reduction rate per unit surface area increased markedly with increasing heat-treatment temperature, and the Cr(VI) reduction and SA degradation rates of the sample calcined at 800 C were approximately 18 and 9 times greater, respectively, than those of the sample calcined at 500
C were approximately 18 and 9 times greater, respectively, than those of the sample calcined at 500 C. Leaching tests of the reaction products revealed that the elution of Ca
C. Leaching tests of the reaction products revealed that the elution of Ca and Cl
and Cl was particularly significant. Even the samples that retained the biotite structure after heat treatment displayed some photocatalytic activity, indicating that this method may be applicable to preparing photocatalysts from mica and related minerals with similar structures.
was particularly significant. Even the samples that retained the biotite structure after heat treatment displayed some photocatalytic activity, indicating that this method may be applicable to preparing photocatalysts from mica and related minerals with similar structures.
吉越 章隆
阿部 真之*; 勝部 大樹*; 大野 真也*
【課題】 効率的なN元素ドープTIO2の製造方法を提供する。 【解決手段】本発明に係るN元素ドープTIO2の製造方法は、ターゲットとなるTIO2を真空チャンバ内の所定位置に配置するステップと、真空チャンバを真空引きするステップと、ノズルを所定温度に加熱するステップと、ノズルを所定温度に維持しつつ、ノズルからNO分子の超音速分子線をターゲットに照射するステップとを備える。
杉田 剛; 下山 巖
not registered
【課題】入手が容易な材料から環境負荷の低い方法で光触媒を製造する方法を提供する。 【解決手段】粘土鉱物と塩とを混合し、500℃以上で加熱することによる、光触媒の製造方法。前記光触媒と金属化合物を混合し、光を照射することによって、前記金属化合物を還元することを特徴とする前記金属化合物の還元方法。